Advanced Energy Sales increased 44% year-over-year in Q4
CLEARWATER, FL — MARCH 31, 2021 – Apyx® Medical Corporation (NASDAQ:APYX) (the “Company”), a maker of medical devices and supplies and the developer of Helium Plasma Technology, marketed and sold as Renuvion® in the cosmetic surgery market and J-Plasma® in the hospital surgical market, today reported financial results for its fourth quarter and full year ended December 31, 2020, and introduced financial expectations for the full year ending December 31, 2021.
Fourth Quarter 2020 Financial Summary:
- Total revenue of $11.5 million, up 36.8% year-over-year.
- Advanced Energy revenue of $9.9 million, up 44.0% year-over-year.
- OEM revenue of $1.6 million, up 4.0% year-over-year.
- Net loss of $1.5 million, compared to net loss of $5.4 million for the fourth quarter of 2019.
- Adjusted EBITDA loss of $0.7 million, compared to adjusted EBITDA loss of $4.8 million for the fourth quarter of 2019.
Full Year 2020 Financial Summary:
- Total revenue of $27.7 million, down 1.9% year-over-year.
- Advanced Energy revenue of $22.2 million, down 2.0% year-over-year.
- OEM revenue of $5.5 million, down 1.1% year-over-year.
- Net loss of $11.9 million, compared to net loss of $19.7 million for 2019.
- Adjusted EBITDA loss of $14.5 million, compared to adjusted EBITDA loss of $16.9 million for 2019.
- As of December 31, 2020, the Company had cash and cash equivalents of $41.9 million, compared to $58.8 million as of December 31, 2019. As of December 31, 2020, the Company had working capital of $56.9 million, including expected cash tax refunds of approximately $7.5 million the Company expects to receive during 2021 related to the net operating loss carrybacks resulting from the 2020 CARES Act.
Fourth Quarter 2020 Highlights:
- On October 20, 2020 the Company announced the publication of two separate peer-reviewed articles published in the journal, Dermatological Reviews:
- “A Retrospective Chart Review of Subdermal Neck Coagulation Using Helium Plasma Technology” (Article link: https://doi.org/10.1002/der2.32)
- “Safety and Efficacy of Helium Plasma for Subdermal Coagulation” (Article link: https://doi.org/10.1002/der2.34)
- On November 9, 2020, the Company announced that it has completed subject enrollment in a U.S. Investigational Device Exemption (IDE) clinical study evaluating the use of its Renuvion technology in dermal resurfacing procedures.
- On November 9, 2020, the Company announced that it received FDA approval for its Safety Report for Feasibility Study (Phase I) of its U.S. IDE clinical study evaluating the use of its Renuvion technology in skin laxity procedures in the neck and submental region.
- On November 23, 2020, the Company announced that it received FDA approval to begin Phase II of its U.S. IDE clinical study evaluating the use of its Renuvion technology in skin laxity procedures in the neck and submental region.
Management Comments:
“Our team brought 2020 to a strong finish by achieving 44% year-over-year revenue growth in the fourth quarter, despite the continued impacts related to the COVID-19 pandemic,” said Charlie Goodwin, President and Chief Executive Officer. “In the fourth quarter, we were pleased to see strong, utilization-based demand in the U.S., with handpiece sales increasing 80%, coupled with 75% year-over-year growth in handpiece sales to our international customers. While demand for our generators in the U.S. continued to be affected by the softer capital equipment purchasing environment due to COVID-19, international generator sales increased 88% year-over-year, driven by demand from several of the new countries that we entered during 2020. In addition to our strong sales performance, we also reported solid margin improvements driven by our efforts to reduce the per unit manufacturing costs of our Advanced Energy handpieces, as well as our focus on reducing discretionary spending in response to the challenging operating environment. Lastly, we delivered important progress with respect to our two U.S. IDE clinical studies, which are designed support our pursuit of new clinical indications for target cosmetic surgery procedures.”
Mr. Goodwin continued: “We introduced full year 2021 financial guidance which calls for revenue growth in the range of 32% to 40% year-over-year, driven by Advanced Energy growth of 45% to 55% year-over-year. Importantly, our guidance assumes that strong handpiece demand from global customers will be the largest contributor to our year-over-year growth, coupled with measured improvement in the capital equipment environment as we move through the year. Our team remains focused on providing unparalleled service and support to our existing customers, while continuing to educate potential new accounts on the benefits of our Renuvion technology via virtual means. We will also continue to advance our long-term strategic initiatives to raise awareness and facilitate the adoption of our Renuvion technology in the cosmetic surgery market. Longer term, we believe Apyx Medical is poised to deliver strong, sustained growth and improving profitability, with an expanded global addressable market opportunity, a strong balance sheet and our truly differentiated Helium Plasma Technology.”
The following tables represent revenue by reportable segment and geography:
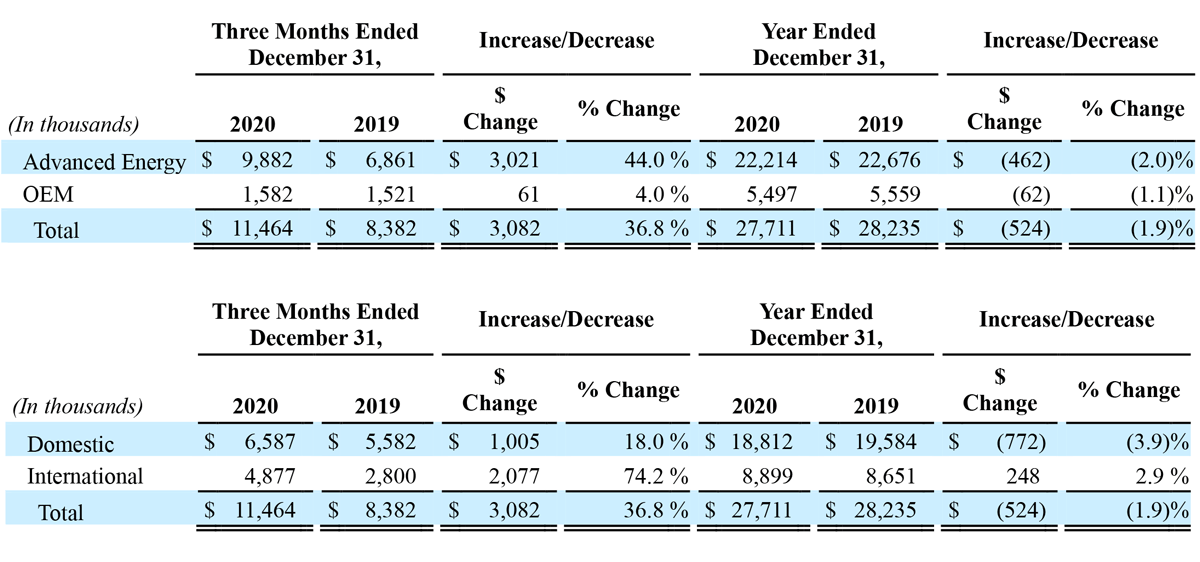
Fourth Quarter 2020 Results:
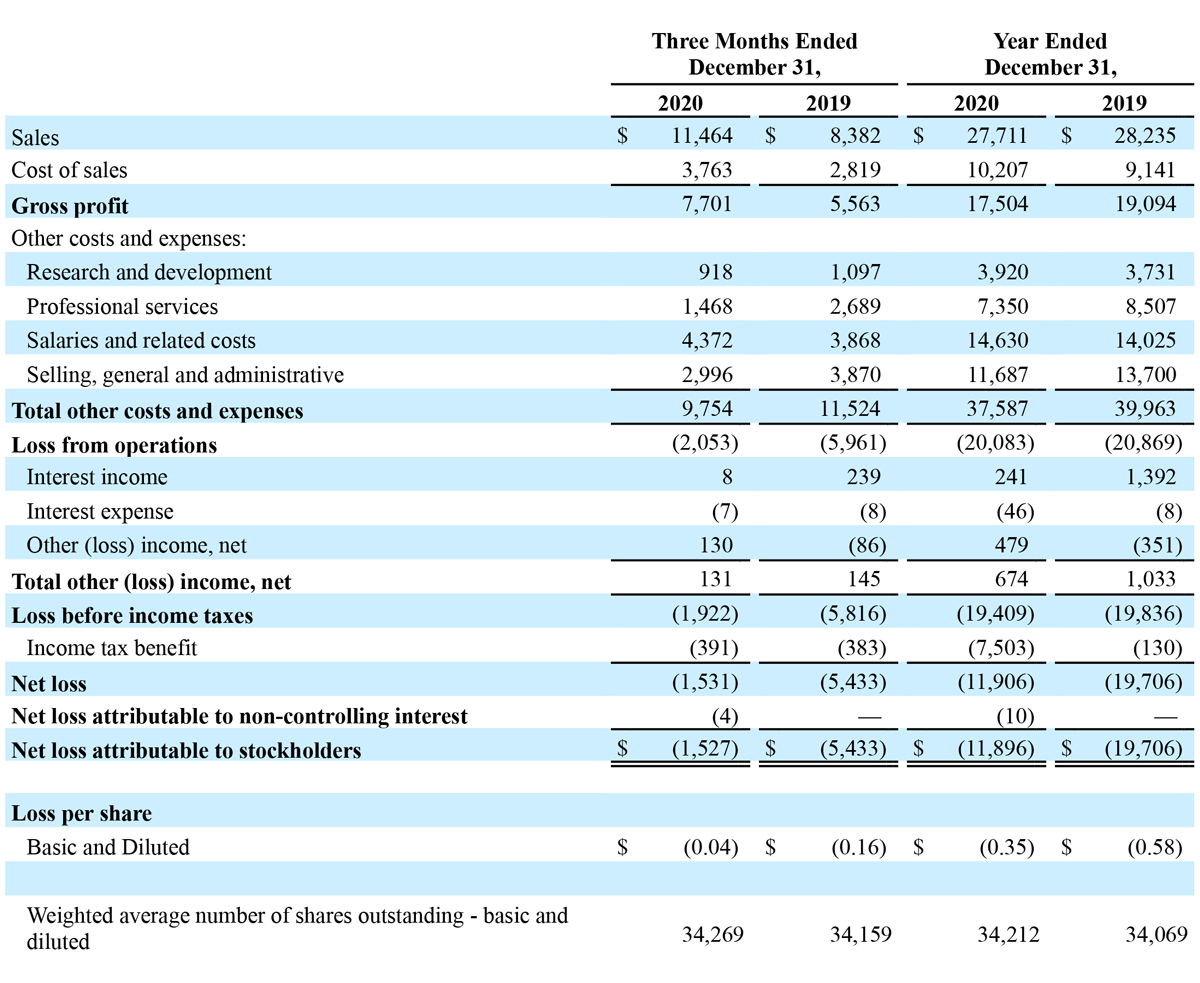
Total revenue for the three months ended December 31, 2020, increased $3.1 million, or 36.8%, to $11.5 million, compared to $8.4 million in the prior year period. Advanced Energy segment sales increased approximately $3.0 million, or 44.0% year-over-year, to $9.9 million, compared to approximately $6.9 million in the prior year period. OEM segment sales increased $0.1 million, or 4.0% year-over-year, to $1.6 million compared to $1.5 million last year. For the fourth quarter 2020, revenue in the United States increased $1.0 million, or 18.0% year-over-year, to $6.6 million, and international revenue increased $2.1 million, or 74.2% year-over-year, to $4.9 million. During the fourth quarter, we saw increased, utilization-based demand for our handpieces domestically, coupled with increased demand for handpieces and generators from our expanded network of international distributors. We continued to see reduced demand for our generators domestically, due to the impact of the COVID-19 pandemic.
Gross profit for the three months ended December 31, 2020, increased $2.1 million, or 38.4% year-over-year, to $7.7 million, compared to $5.6 million in the prior year period. Gross margin for the three months ended December 31, 2020, was 67.2%, compared to 66.4% for the same period in 2019. The increase in profit margins for the three months ended December 31, 2020 from the prior year period is primarily attributable to product mix within our Advanced Energy segment, as we experienced increased demand for generators internationally, and improved product margins in our Advanced Energy segment as a result of our continued manufacturing efficiency initiatives and the introduction of newer product models. The increased profit margins was partially offset by product mix within the OEM segment and geographical revenue mix due to international sales growth outpacing domestic sales growth.
Operating expenses for the fourth quarter of 2020 decreased $1.8 million, or 15.4% year-over-year, to $9.8 million, compared to $11.5 million for the fourth quarter of 2019. The year-over-year change in operating expenses was driven by a $1.2 million decrease in professional services, a $0.9 million decrease in selling, general and administrative expenses, and a $0.2 million decrease in research and development expenses. The year-over-year change in operating expenses was partially offset by a $0.5 million increase in salaries and related costs.
Net loss for fourth quarter of 2020 was $1.5 million, or $(0.04) per share, compared to a net loss of $5.4 million, or $(0.16) per share, for the fourth quarter of 2019. The year-over-year decrease in net loss was primarily due to the increase in gross profit of $2.1 million and the decrease in operating expenses of $1.8 million.
Full Year 2020 Results:
Total revenue for the year ended December 31, 2020, decreased $0.5 million, or 1.9%, to $27.7 million, compared to $28.2 million in the prior year. Advanced Energy segment sales decreased $0.5 million, or 2.0% year-over-year, to $22.2 million, compared to $22.7 million last year. OEM segment sales decreased $0.1 million, or 1.1% year-over-year, to $5.5 million, compared to $5.6 million last year. For the year ended December 31, 2020, revenue in the United States decreased $0.8 million, or 3.9% year-over-year, to $18.8 million, while international revenue increased $0.2 million, or 2.9% year-over-year, to $8.9 million. The impact of COVID-19 resulted in decreased demand for our products, both domestically and internationally throughout 2020, although, sales began to recover late in the second quarter, and through the end of the year, as many of our customers resumed operations in a limited capacity. Demand for our products internationally benefited from our entry into multiple new markets during 2020, the largest of which was Brazil.
Net loss for the year ended December 31, 2020 was $11.9 million, or $(0.35) per share, compared to a net loss of $19.7 million, or $(0.58) per share, for the year ended December 31, 2019. The year-over-year decrease in net loss for 2020 was primarily due to the increase in the Company’s income tax benefit of $7.4 million related to its provision for the carryback of net operating losses for 2019 and 2020, and the decrease in operating expenses of $2.4 million. The year-over-year decrease in net loss was offset partially by the year-over-year decrease in gross profit and other income of $1.6 million and $0.4 million, respectively.
Full Year 2021 Financial Outlook:
The Company is introducing financial guidance for the year ending December 31, 2021 of:
- Total revenue in the range of $36.7 million to $38.7 million, representing growth of 32% to 40% year-over-year, compared to total revenue of $27.7 million for the year ended December 31, 2020.
- Total revenue guidance assumes:
- Advanced Energy revenue in the range of approximately $32.3 million to $34.3 million, representing growth of 45% to 55% year-over-year, compared to Advanced Energy revenue of $22.2 million for the year ended December 31, 2020. The Advanced Energy revenue range assumes that U.S. growth is only driven by contributions from Renuvion sales related to its use as a sub-dermal coagulator following liposuction procedures and that international growth is driven primarily by demand in existing international markets.
- OEM revenue of approximately $4.4 million, representing a decline of 20% year-over-year, compared to $5.5 million for the year ended December 31, 2020.
- Total revenue guidance assumes:
- Net loss attributable to stockholders in the range of $20.7 million to $18.4 million, compared to net loss attributable to stockholders of $11.9 million for the year ended December 31, 2020.
- Adjusted EBITDA loss in the range of $14.6 million to $12.0 million, compared to adjusted EBITDA loss of $14.5 million for the year ended December 31, 2020.
Conference Call Details:
Management will host a conference call at 8:00 a.m. Eastern Time on March 31 to discuss the results of the quarter and to host a question and answer session. To listen to the call by phone, interested parties may dial 877-407-8289 (or 201-689-8341 for international callers) and provide access code 13715804. Participants should ask for the Apyx Medical Corporation Call. A live webcast of the call will be accessible via the Investor Relations section of the Company’s website and at:
https://78449.themediaframe.com/dataconf/productusers/apyx/mediaframe/43349/indexl.html
A telephonic replay will be available approximately two hours after the end of the call through April 14, 2021. The replay can be accessed by dialing 877-660-6853 for U.S. callers or 201-612-7415 for international callers and using the replay access code: 13715804 The webcast will be archived on the Investor Relations section of the Company’s website.
Investor Relations Contact:
Westwicke Partners on behalf of Apyx Medical Corporation
Mike Piccinino, CFA
investor.relations@apyxmedical.com
About Apyx Medical Corporation:
Apyx Medical Corporation is an advanced energy technology company with a passion for elevating people’s lives through innovative products in the cosmetic and surgical markets. Known for its innovative Helium Plasma Technology, Apyx is solely focused on bringing transformative solutions to the physicians and patients it serves. The Company’s Helium Plasma Technology is marketed and sold as Renuvion in the cosmetic surgery market and J-Plasma in the hospital surgical market. Renuvion offers surgeons and physicians a unique ability to provide controlled heat to the tissue to achieve their desired results. The J-Plasma system allows surgeons to operate with a high level of precision and virtually eliminating unintended tissue trauma. The Company also leverages its deep expertise and decades of experience in unique waveforms through original equipment manufacturing (OEM) agreements with other medical device manufacturers. For further information about the Company and its products, please refer to the Apyx Medical Corporation website at www.ApyxMedical.com.
Cautionary Statement on Forward-Looking Statements:
Certain matters discussed in this release and oral statements made from time to time by representatives of the Company may constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and the Federal securities laws. Although the Company believes that the expectations reflected in such forward-looking statements are based upon reasonable assumptions, it can give no assurance that its expectations will be achieved.
Forward-looking information is subject to certain risks, trends and uncertainties that could cause actual results to differ materially from those projected. Many of these factors are beyond the Company’s ability to control or predict. Important factors that may cause actual results to differ materially and that could impact the Company and the statements contained in this release can be found in the Company’s filings with the Securities and Exchange Commission including the Company’s Report on Form 10-K for the year ended December 31, 2020. For forward-looking statements in this release, the Company claims the protection of the safe harbor for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995. The Company assumes no obligation to update or supplement any forward-looking statements whether as a result of new information, future events or otherwise.
APYX MEDICAL CORPORATION
CONSOLIDATED STATEMENTS OF OPERATIONS
(In thousands, except per share data)
APYX MEDICAL CORPORATION
CONSOLIDATED BALANCE SHEETS
(In thousands, except share and per share data)

APYX MEDICAL CORPORATION
RECONCILIATION OF GAAP NET LOSS RESULTS TO NON-GAAP ADJUSTED EBITDA
(Unaudited)
Use of Non-GAAP Financial Measure
We present the following non-GAAP measure because we believe such measure is a useful indicator of our operating performance. Our management uses this non-GAAP measure principally as a measure of our operating performance and believes that this measure is useful to investors because it is frequently used by analysts, investors and other interested parties to evaluate companies in our industry. We also believe that this measure is useful to our management and investors as a measure of comparative operating performance from period to period.
The Company has presented the following non-GAAP financial measure in this press release: adjusted EBITDA. The Company defines adjusted EBITDA as its reported net income/(loss) attributable to stockholders (GAAP) plus income tax expense (benefit), interest, depreciation and amortization, and stock-based compensation expense.

The following unaudited table presents a reconciliation of net loss to Adjusted EBITDA for the year ending December 31, 2021. The reconciliation assumes the mid-point of the Adjusted EBITDA loss range and the midpoint of each component of the reconciliation, corresponding to guidance for GAAP net loss attributable to stockholders of $20.7 million to $18.4 million for the year ending December 31, 2021.

