Glossary of Symbols
| Standard Reference | Symbol | Symbol Title | Explanatory Text |
|---|---|---|---|
ISO 15223-1 |
|||
| 5.1.1 |  |
Manufacturer | Indicates the medical device manufacturer. |
| 5.1.2 | Authorized representative in the European Community | Indicates the authorized representative in the European Community / European Union. | |
| 5.1.3 | 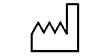 |
Date of manufacture | Indicates the date when the medical device was manufactured |
| 5.1.4 | 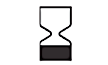 |
Use-by date | Indicates the date after which the medical device is not to be used. |
| 5.1.5 | Batch code | Indicates the manufacturer’s batch code so that the batch or lot can be identified. | |
| 5.1.6 |  |
Catalogue number | Indicates the manufacturer’s catalog number so that the medical device can be identified. |
| 5.2.3 | Sterilized using ethylene oxide | Indicates a medical device that has been sterilized using ethylene oxide. | |
| 5.2.4 | Sterilized using irradiation | Indicates a medical device that has been sterilized using irradiation.. | |
| 5.2.6 | Do not resterilize | Indicates a medical device that is not to be resterilized | |
| 5.2.7 |  |
Non-sterile | Indicates a medical device that has not been subjected to a sterilization process. |
| 5.2.8 |  |
Do not use if package is damaged | Indicates a medical device that should not be used if the package has been damaged or opened and that the user should consult the instructions for use for additional information |
| 5.2.11 |  |
Single Sterile barrier system | Indicates a single sterile barrier system. |
| 5.2.14 |  |
Single sterile barrier system with protective packaging outside | Indicates a single sterile barrier system with protective packaging outside. |
| 5.3.7 | 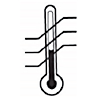 |
Temperature Limit | Indicates the temperature limits to which the medical device can be safely exposed. |
| 5.3.8 | 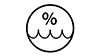 |
Humidity Limitation | Indicates the range of humidity to which the medical device can be safely exposed. |
| 5.4.2 | 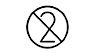 |
Do not re-use | Indicates a medical device that is intended for one single use only. NOTE: Synonyms for “Do not reuse” are “single use” and “use only once”. |
| 5.4.3 | Consult instructions for use | Indicates the need for the user to consult the instructions for use. | |
| 5.4.4 |  |
Caution | To indicate that caution is necessary when operating the device or control close to where the symbol is placed, or to indicate that the current situation needs operator awareness or operator action in order to avoid undesirable consequences. |
FDAUse of Symbols |
Prescription Use Only | Caution: Federal law (USA) restricts this device to sale by or on the order of a licensed healthcare practitioner. | |
EU MDR 2017/745 |
Medical Device | Indicates the device is a medical device. | |
DIRECTIVE 2012/19/EUAnnex II |
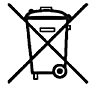 |
WEEE Label | Indicates that the product should not be discarded as unsorted waste but must be sent to separate collection facilities for recovery and recycling. |
ASTM F25037.3.3 |
MR Unsafe | An item which poses unacceptable risks to the patient, medical staff or other persons within the MR environment. | |
ABBREVIATIONS |
Qty: | Quantity | Quantity of devices in package. |
IEC 60417 |
 |
Alternating Current | |
IEC 60417 |
 |
Type BF Class I | |
IEC 60417 |
 |
Fuse |

