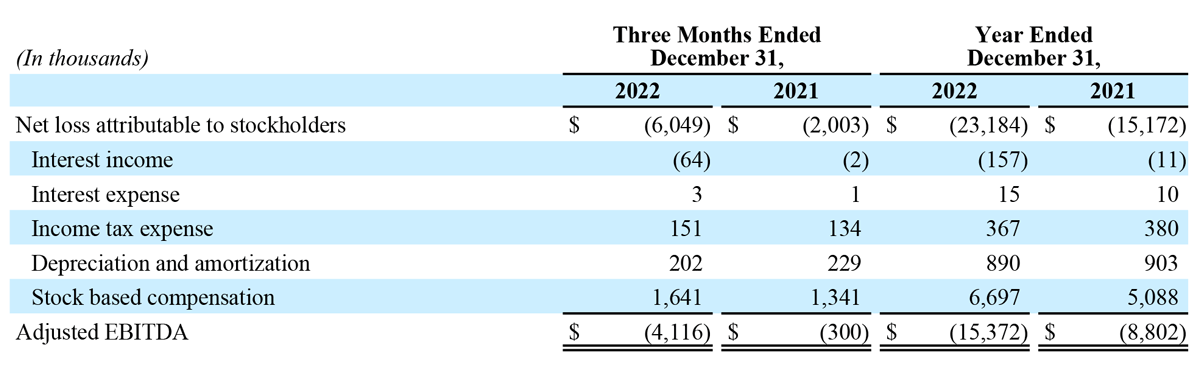APYX MEDICAL CORPORATION REPORTS FOURTH QUARTER AND FULL YEAR 2022 FINANCIAL RESULTS; INTRODUCES FULL YEAR 2023 FINANCIAL OUTLOOK
CLEARWATER, FL — March 16, 2023 – Apyx® Medical Corporation (NASDAQ:APYX) (the “Company”), the manufacturer of a proprietary helium plasma and radiofrequency technology marketed and sold as Renuvion®, today reported financial results for its fourth quarter and full year ended December 31, 2022, and introduced financial expectations for the full year ending December 31, 2023.
Fourth Quarter 2022 Financial Summary:
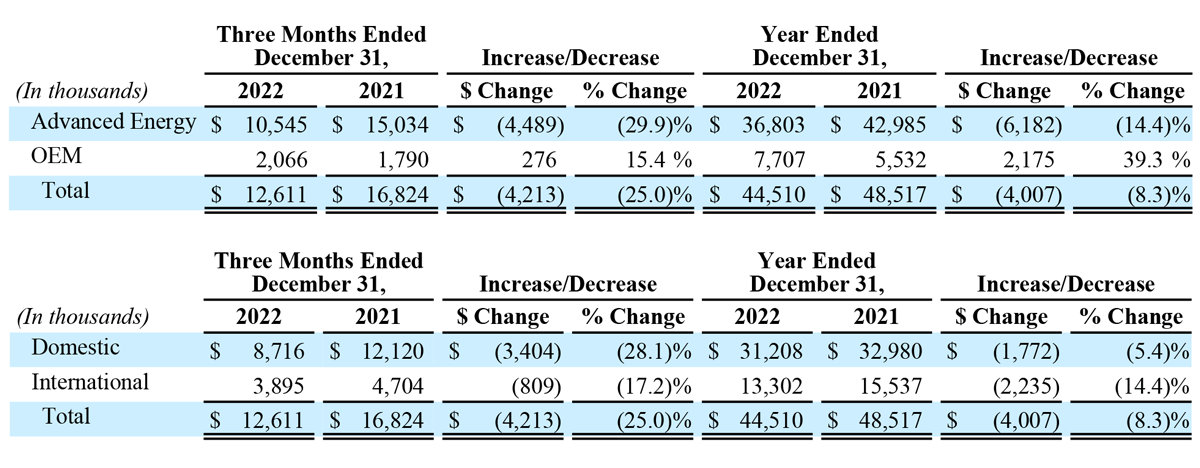
- Total revenue of $12.6 million, down 25% year-over-year.
- Advanced Energy revenue of $10.5 million, down 30% year-over-year.
- OEM revenue of $2.1 million, up 15% year-over-year.
- Net loss attributable to stockholders of $6.0 million, compared to $2.0 million for the fourth quarter of 2021.
- Adjusted EBITDA loss of $4.1 million, compared to $0.3 million for the fourth quarter of 2021.
Full Year 2022 Financial Summary:
- Total revenue of $44.5 million, down 8% year-over-year.
- Advanced Energy revenue of $36.8 million, down 14% year-over-year.
- OEM revenue of $7.7 million, up 39% year-over-year.
- Net loss attributable to stockholders of $23.2 million, compared to $15.2 million for 2021.
- Adjusted EBITDA loss of $15.4 million, compared to $8.8 million for 2021.
Highlights & Developments Subsequent to Quarter End:
- On January 3, 2023, the Company announced the launch of its first direct-to-consumer brand campaign. Entitled #ThisIsMe, the campaign is aimed at U.S. consumers who are interested in a minimally invasive procedure with the Renuvion technology.
- On January 25, 2023, the Company announced the launch of its latest-generation Renuvion generator, the Apyx One Console, in the United States.
- On February 1, 2023, the Company announced it submitted a 510(k) premarket notification (“510(k) submission”) for the Renuvion APR Handpiece to the U.S. Food and Drug Administration (“FDA”), supported by a clinical study and real-world evidence. The 510(k) submission is intended to expand Renuvion’s indications for use to include a specific indication for the use of the Renuvion APR Handpiece for the coagulation of subcutaneous soft tissues where needed, following liposuction.
- On February 21, 2023, the Company announced that it and its subsidiaries had entered into a new, five-year secured credit facility with MidCap Financial. The credit agreement provides for an up to $35 million facility consisting of senior, secured term loans of up to $25 million and a revolving facility of up to $10 million.
- On February 27, 2023, the Company announced that it received 510(k) clearance from the FDA “for the use of the Renuvion APR Handpiece for the delivery of radiofrequency energy and/or helium plasma where coagulation/contraction of soft tissue is needed. Soft tissue includes subcutaneous tissue.”
Management Comments:
“Our revenue performance in the fourth quarter reflected the continued business disruption related to the Medical Device Safety Communication,” said Charlie Goodwin, President and Chief Executive Officer. “While Advanced Energy revenue in the fourth quarter increased 49% sequentially, our results were ultimately below our expectations as global sales of our Advanced Energy generators remained lower than anticipated. In spite of these challenges, our team remained focused on educating the market about the strong safety and efficacy profile of our products and the additional 510(k) clearances we secured in 2022. In tandem, we continued to pursue regulatory clearance for a specific clinical indication which we believe will directly address the remaining limitations of the Safety Communication. To this end, we engaged with the FDA to obtain and incorporate their feedback on our proposed regulatory submission for this indication during the fourth quarter, and submitted our request for 510(k) clearance at the end of January.”
Mr. Goodwin continued: “While 2022 ultimately proved to be a challenging year, our team made important progress under difficult circumstances. We believe we are incrementally better positioned in 2023, with an expanding portfolio of 510(k) clearances for our targeted clinical indications, recently implemented activities to reduce operating expenses while preserving our capabilities as an organization, and additional financing secured to strengthen our balance sheet and enhance our financial flexibility. In 2023, we expect to grow global sales of our Advanced Energy products in excess of 35%, fueled by strong execution on our commercial and regulatory objectives, improving generator demand and handpiece utilization trends as we move through fiscal 2023. With large and rapidly growing addressable markets, unique technology supported by a strong portfolio of clinical evidence, and a dedicated team of employees, we are excited about our future prospects in 2023 and the years to come.”
The following tables present revenue by reportable segment and geography:
Fourth Quarter 2022 Results:
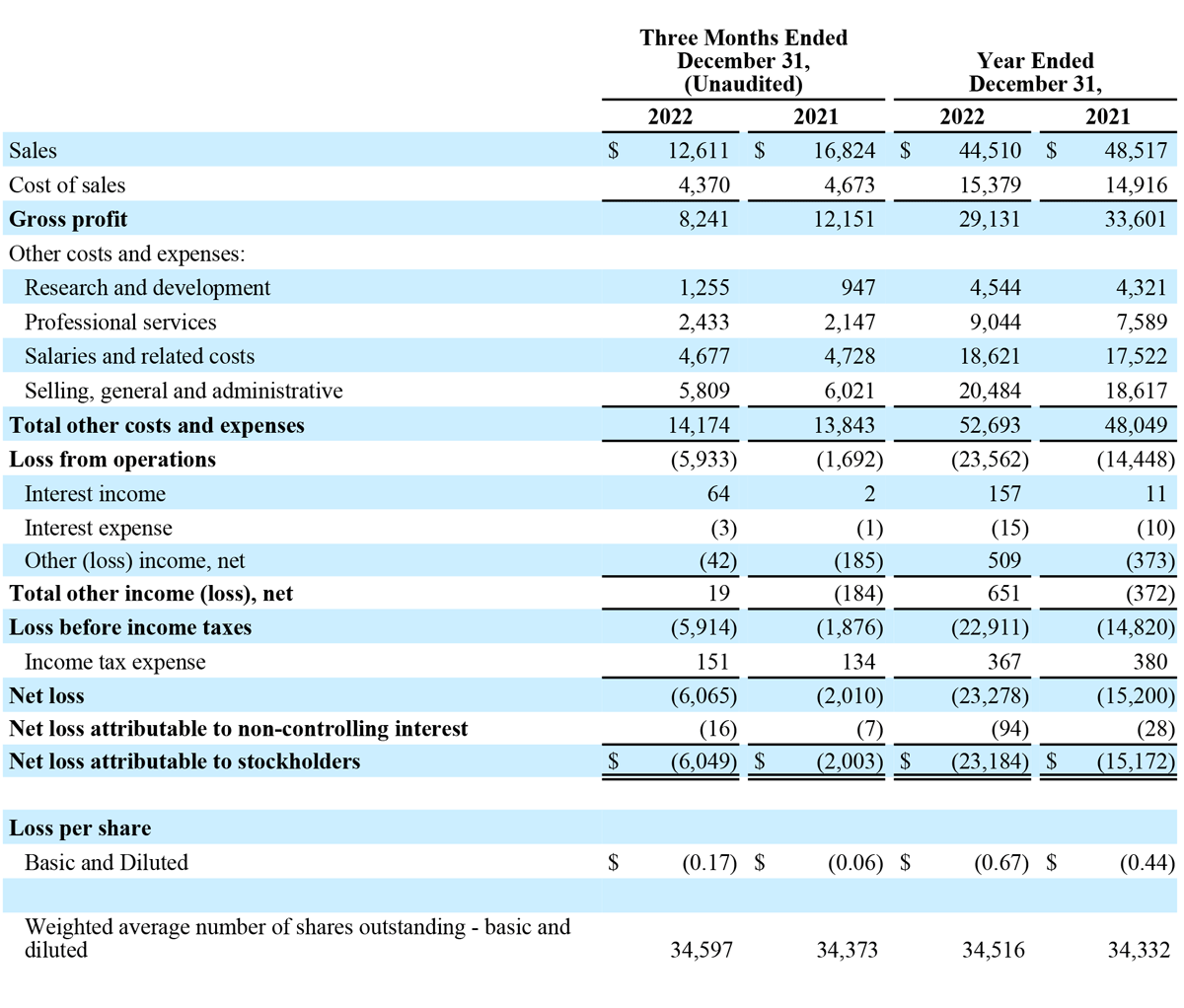
Total revenue for the three months ended December 31, 2022 decreased $4.2 million, or 25% year-over-year, to $12.6 million, compared to $16.8 million in the prior year period. Advanced Energy segment sales decreased $4.5 million, or 30% year-over-year, to $10.5 million. OEM segment sales increased $0.3 million, or 15% year-over-year to $2.1 million. The year-over-year decrease in Advanced Energy revenue was due to decreased global demand for the Company’s handpieces and generators following the FDA Safety Communication on March 14, 2022. The increase in OEM sales was due to increases in sales volume to existing customers, including Symmetry Surgical, under our 10-year generator manufacturing and supply agreement, as well as incremental new sales upon the commencement of the supply arrangement related to the completion of the development portion of some of our OEM development agreements. For the fourth quarter of 2022, revenue in the United States decreased $3.4 million, or 28% year-over-year, to $8.7 million, and international revenue decreased $0.8 million, or 17% year-over-year, to $3.9 million.
Gross profit for the three months ended December 31, 2022, decreased $3.9 million, or 32% year-over-year, to $8.2 million. Gross margin for the three months ended December 31, 2022, was 65.3%, compared to 72.2% in the prior year period. The decrease in gross profit margins for the three months ended December 31, 2022 from the prior year period was primarily attributable to changes in the sales mix between our two segments, product mix within our Advanced Energy Segment and higher material and inbound shipping costs to manufacture our inventory. These decreases were partially offset by geographic mix within our Advanced Energy segment, with higher margin direct domestic sales comprising a higher percentage of total sales, and by the increased mix of newer product models.
Operating expenses for the three months ended December 31, 2022 increased $0.3 million, or 2% year-over-year, to $14.2 million. The year-over-year change in operating expenses was driven by a $0.3 million increase in research and development and a $0.3 million increase in professional services. These increases were partially offset by a $0.2 million decrease in selling, general and administrative expenses and a $0.1 million decrease in salaries and related costs.
Income tax expense for the three months ended December 31, 2022 and 2021 was $0.2 million and $0.1 million, respectively.
Net loss attributable to stockholders for the three months ended December 31, 2022 was $6.0 million, or $0.17 per share, compared to $2.0 million, or $0.06 per share, in the prior year period.
Adjusted EBITDA loss for the three months ended December 31, 2022 was $4.1 million, compared to $0.3 million in the prior year period.
Full Year 2022 Results:
Total revenue for the year ended December 31, 2022, decreased $4.0 million, or 8%, to $44.5 million. Advanced Energy segment sales decreased $6.2 million, or 14% year-over-year, to $36.8 million. OEM segment sales increased $2.2 million, or 39% year-over-year, to $7.7 million. The year-over-year decrease in Advanced Energy revenue was due to decreased global demand for the Company’s handpieces and generators following the FDA Safety Communication on March 14, 2022. The increase in OEM sales was due to increases in sales volume to existing customers, including Symmetry Surgical, under our 10-year generator manufacturing and supply agreement, as well as incremental new sales upon the commencement of the supply arrangement related to the completion of the development portion of some of our OEM development agreements. For 2022, revenue in the United States decreased $1.8 million, or 5% year-over-year, to $31.2 million, and international revenue decreased $2.2 million, or 14% year-over-year, to $13.3 million.
Net loss attributable to stockholders for the year ended December 31, 2022 was $23.2 million, or $0.67 per share, compared to $15.2 million, or $0.44 per share, in the prior year period.
Full Year 2023 Financial Outlook:
The Company is introducing financial guidance for the year ending December 31, 2023 to:
- Total revenue in the range of $58.0 million to $61.0 million, representing growth of approximately 30% to 37% year-over-year, compared to total revenue of $44.5 million for the year ended December 31, 2022.
- Total revenue guidance assumes:
- Advanced Energy revenue in the range of $50.0 million to $53.0 million, representing growth of approximately 36% to 44% year-over-year, compared to Advanced Energy revenue of $36.8 million for the year ended December 31, 2022.
- OEM revenue of approximately $8 million, representing growth of approximately 4% year-over-year, compared to $7.7 million for the year ended December 31, 2022.
- Total revenue guidance assumes:
- Net loss attributable to stockholders of approximately $14.0 million, compared to $23.2 million for the year ended December 31, 2022.
Conference Call Details:
Management will host a conference call at 8:00 a.m. Eastern Time on March 16, 2023 to discuss the results of the quarter and year, and to host a question and answer session. To listen to the call by phone, interested parties may dial 877-407-8289 (or 201-689-8341 for international callers) and provide access code 13736530. Participants should ask for the Apyx Medical Corporation Call. A live webcast of the call will be accessible via the Investor Relations section of the Company’s website and at:
https://event.choruscall.com/mediaframe/webcast.html?webcastid=RDPQw6xg
A telephonic replay will be available approximately two hours after the end of the call through the following two weeks. The replay can be accessed by dialing 877-660-6853 for U.S. callers or 201-612-7415 for international callers and using the replay access code: 13736530. The webcast will be archived on the Investor Relations section of the Company’s website.
Investor Relations Contact:
ICR Westwicke on behalf of Apyx Medical Corporation
Mike Piccinino, CFA
investor.relations@apyxmedical.com
About Apyx Medical Corporation:
Apyx Medical Corporation is an advanced energy technology company with a passion for elevating people’s lives through innovative products, including its Helium Plasma Technology products marketed and sold as Renuvion in the cosmetic surgery market and J-Plasma® in the hospital surgical market. Renuvion and J-Plasma offer surgeons a unique ability to provide controlled heat to tissue to achieve their desired results. The Company also leverages its deep expertise and decades of experience in unique waveforms through OEM agreements with other medical device manufacturers. For further information about the Company and its products, please refer to the Apyx Medical Corporation website at www.ApyxMedical.com.
Cautionary Statement on Forward-Looking Statements:
Certain matters discussed in this release and oral statements made from time to time by representatives of the Company may constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and the Federal securities laws. Although the Company believes that the expectations reflected in such forward-looking statements are based upon reasonable assumptions, it can give no assurance that its expectations will be achieved.
All statements other than statements of historical fact are statements that could be deemed forward-looking statements, including but not limited to, any statements regarding the potential impact of the COVID-19 pandemic and the actions by governments, businesses and individuals in response to the situation; projections of net revenue, margins, expenses, net earnings, net earnings per share, or other financial items; projections or assumptions concerning the possible receipt by the Company of any regulatory approvals from any government agency or instrumentality including but not limited to the U.S. Food and Drug Administration, supply chain disruptions, component shortages, manufacturing disruptions or logistics challenges; or macroeconomic or geopolitical matters and the impact of those matters on the Company’s financial performance.
Forward-looking statements and information are subject to certain risks, trends and uncertainties that could cause actual results to differ materially from those projected. Many of these factors are beyond the Company’s ability to control or predict. Important factors that may cause the Company’s actual results to differ materially and that could impact the Company and the statements contained in this release include but are not limited to risks, uncertainties and assumptions relating to the regulatory environment in which the Company is subject to, including the Company’s ability to gain requisite approvals for its products from the U.S. Food and Drug Administration and other governmental and regulatory bodies, both domestically and internationally; the impact of the recent FDA Safety Communication on our business and operations; factors relating to the effects of the COVID-19 pandemic; sudden or extreme volatility in commodity prices and availability, including supply chain disruptions; changes in general economic, business or demographic conditions or trends; changes in and effects of the geopolitical environment; liabilities and costs which the Company may incur from pending or threatened litigations, claims, disputes or investigations; and other risks that are described in the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2022 and the Company’s other filings with the Securities and Exchange Commission. For forward-looking statements in this release, the Company claims the protection of the safe harbor for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995. The Company assumes no obligation to update or supplement any forward-looking statements whether as a result of new information, future events or otherwise.
APYX MEDICAL CORPORATION
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS
(In thousands, except per share data)
APYX MEDICAL CORPORATION
CONDENSED CONSOLIDATED BALANCE SHEETS
(In thousands, except share and per share data)
APYX MEDICAL CORPORATION
RECONCILIATION OF GAAP NET LOSS RESULTS TO NON-GAAP ADJUSTED EBITDA
(Unaudited)
Use of Non-GAAP Financial Measure
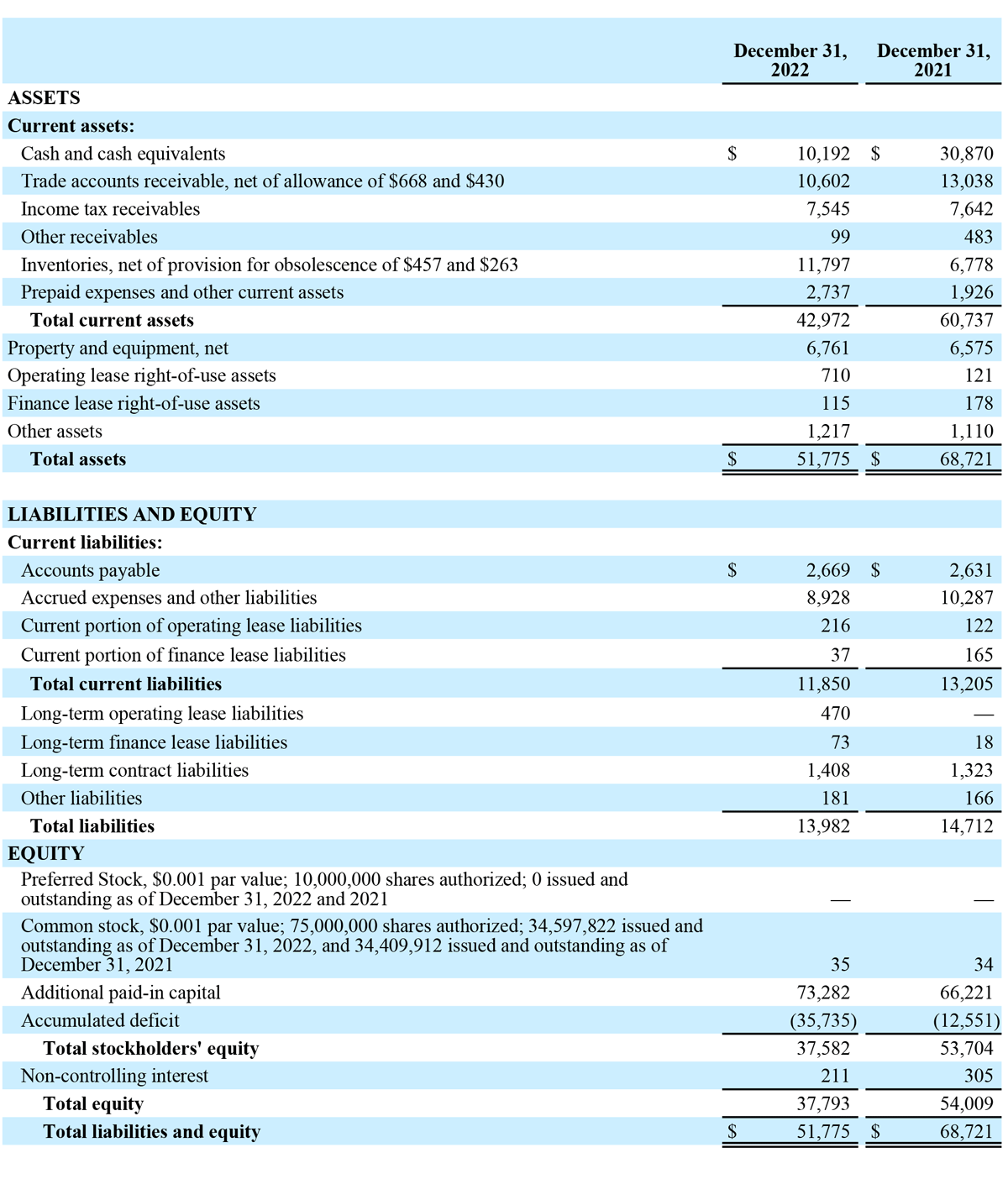
We present the following non-GAAP measure because we believe such measure is a useful indicator of our operating performance. Our management uses this non-GAAP measure principally as a measure of our operating performance and believes that this measure is useful to investors because it is frequently used by analysts, investors and other interested parties to evaluate companies in our industry. We also believe that this measure is useful to our management and investors as a measure of comparative operating performance from period to period. The non-GAAP financial measure presented in this release should not be considered as a substitute for, or preferable to, the measures of financial performance prepared in accordance with GAAP.
The Company has presented the following non-GAAP financial measure in this press release: adjusted EBITDA. The Company defines adjusted EBITDA as its reported net income (loss) attributable to stockholders (GAAP) plus income tax expense (benefit), interest, depreciation and amortization, and stock-based compensation expense.